As per the Grand View Research, the smart medical devices market size is expected to reach USD 24.46 B by 2025. In today’s new-age technology era, smart and effective medical devices play a vital role in human life. Medical devices are the key solutions to healthcare problems that help save human lives faster from serious injuries/diseases and suitably cures human health by boosting up the immunity balance with proper medications and treatments. As it costs life-saving, it’s important to validate the quality of such medical devices before post-market release for public use.
The healthcare organization or medical device manufacturers should follow the common principles for the validation of medical devices defined by the Food and Drug Administration (FDA). The validation process of healthcare devices can help in identifying whether such devices are legal, safe and effective for medical use. Furthermore, the customers’ trust and satisfaction can also be achieved by following such regulated validation processes for medical devices.
Importance of Healthcare Device Testing
Medical device testing is a process that validates the functionality, performance, interoperability, risk factor to patients and safety measurements for medical devices during the design and development stage. It helps to identify whether the requirements of the medical devices are fulfilled as defined and also determines the quality level in medical devices for its continuous improvement.
The involvement of the Quality Assurance (QA) from the beginning can help to reduce the production costs of manufacturing the accurate medical devices by finding defects at earlier stages and can save re-work time of the development team as well.
Identify the Healthcare Device Type before Testing
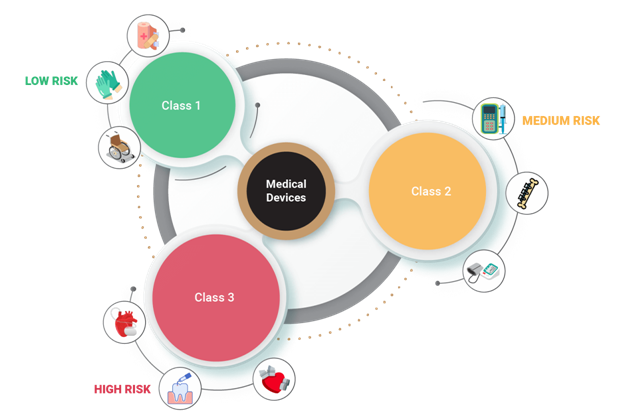
The Food and Drug Administration (FDA) has defined three classifications for all types of healthcare devices. Each of these medical devices is assigned to one of three regulatory classes by FDA based on device risks, health safety measurement controls, effectiveness of the device and impact on the patient’s health. Before testing a medical device as an individual, it’s significant to identify the device type/class as defined by FDA and execute the suitable Test Approach accordingly.
The three classes of medical devices are as follows:
QA Test Approaches to Perform Medical Device Testing
- Gain the knowledge of Quality Standards for Medical Devices:- There are some quality standards/compliances defined globally to ensure the quality of medical devices and are followed before they are offered to patients and healthcare professionals. A QA must learn about those standards followed at each stage of the medical device lifecycleThe quality standards to be followed for medical devices development vary from one application to another, the most common ones being ISO 9001, 13485 for quality management, ISO 14971 for risk management and IEC 62304 for software lifecycle
- Requirements Analysis:- The requirement analysis process plays a major role to identify and understand the testing requirements for a medical device. As it’s the foundation of the Test Plan, QA must understand all the requirements of various categories like software development, UI design, system production, etc. which completes the medical device operations. For the same, QA should be involved early in the project ensuring that issues are identified before they become defects, before the completion of the technical design. This, in turn, saves considerable time and money
- Documentation:- After requirement analysis, the next step is to prepare documents for reference information. The benefit of documentation is that the QA team can effectively determine the quality of medical devices with such detailed information. This also works like the quality metrics for the design and development of medical devices in the form of continuous quality improvement. Following documentation is required: Test plans – Software assurance quality plan and risk management plan
- Test case identification for the features
- Linking the requirements with each test case for the traceability
- Reports of the test cases (pass and fail results) using test management tool like IBM Rational Quality Manager (RQM)
- Environment Test Setup & Sanity Check:- Before initiating the testing process of the medical device, the environmental setup is the most crucial aspect for it. The unavailability or faulty environment setup can ruin all the testing efforts for the medical device. Thus, the environment setup requirements like software and hardware for the medical device testing should be analyzed and arranged accordingly. After preparing the environment setup, QA must perform the sanity check on the key features of the medical device to determine that basic functionalities of the medical device are working as expected and further detailed testing can be processed accordingly
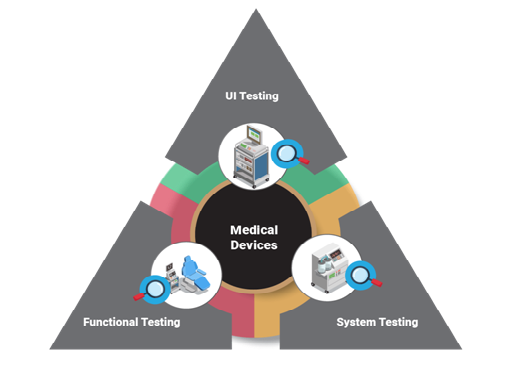
- Software Testing:- The software testing is divided into three parts as follows:
- UI testing: It helps to determine the actual workflow of the medical device by interacting with functions and features
- Functional testing: Also, known as black box testing, its main purpose is to check all the functional requirement of the medical device software being met as per the customer’s development objective
- System testing: It is a testing process where the medical device software is merged with hardware to ensure that both will work as a complete system
- Out of Box Testing (OOB Testing):- The final step of the Test Execution cycle is Out Of Box testing. It is performed on the medical devices to identify whether the device can handle the exceptions and errors caused by environmental, electrical or mechanical factors. In such type of testing, the invalid inputs or a combination of inputs is provided to the medical device to observe its behavior and determine the possible exceptions & errors
The main purpose of out of box testing is to verify that the medical device should not fail to operate its basic functionalities despite such exceptions and errors occurring. It also confirms to what extent the device can effectively handle such exceptions in the future
The above mentioned testing process can also be automated if the medical device to be tested supports testing automation. Automation testing can be performed with any programming language like Python, Robot framework, Ruby, etc. based on the device testing requirements. The main advantage of automation testing is that it reduces the testing efforts and execution time of repetitive tasks by automating the manual testing process
Benefits of Medical Device Testing
- Enhanced reliability of the medical devices with efficient performance
- Ensured compliance with government regulations and ISO standards
- Decreased failure rates, uncertain events and corrective actions, minimizing manufacturer’s liability
- Reduced long term cost involved in the maintenance

About the Author: Shraddha Shah
Shraddha is associated with VOLANSYS Technologies as a QA Automation Lead for more than 2 years. She has vast experience in Web, Mobile and Cloud Automation testing domains.









