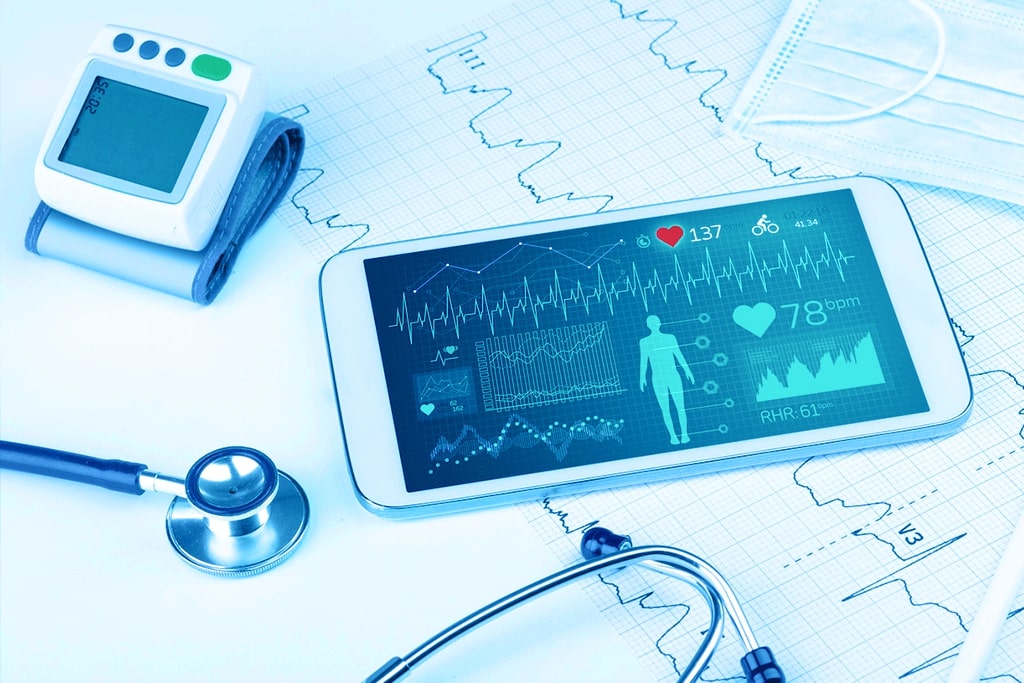
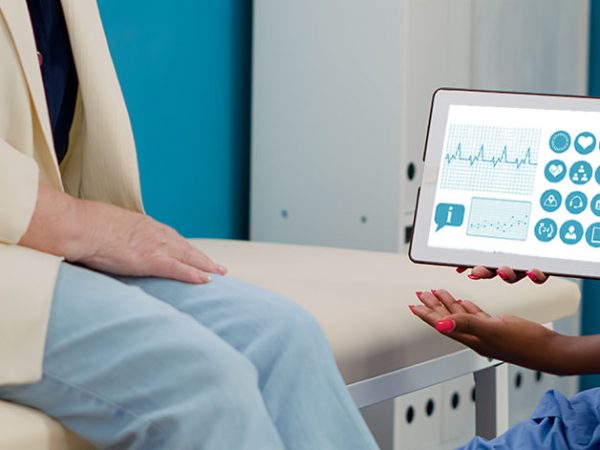
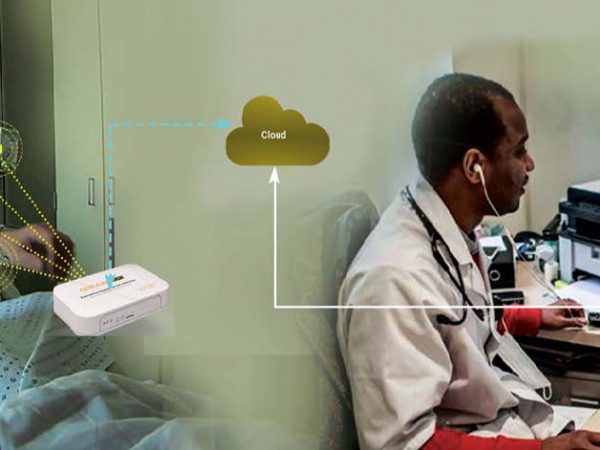
Digital transformation has changed the way the healthcare industry operates. Connected medical devices and remote health monitoring have boosted the precision, efficiency, and reliability of medical treatment. IoT solutions in healthcare have benefited patients, doctors, and hospitals by enabling real-time, remote access to a patient’s vital analysis and health data. Drug delivery systems are moving towards IoT enabled pumps, inhalers, auto-injectors as well as insideables for diagnostic and body function monitoring.
VOLANSYS is a trusted healthcare solution partner for leading medical device manufacturers and healthcare system providers. We have hands-on experience in providing smart healthcare solutions and medical-grade FDA/HIPAA compliant solutions including medical diagnostic devices, robotic surgical equipment, DNA analysis devices, drug delivery systems, and digital health systems using the latest technologies like Wi-Fi, LTE/4G/5G. Our capabilities include device miniaturization, designing low-power customized wearables, invasive and non-invasive devices, security compliance, and more.
Delivering Smart
Healthcare Solutions
IoT Adoption
Medical Regulation Compliance
Remote Medical Assistance
Patient Vital Analysis
Medical/Healthcare
Offerings
Health Devices & Wearables
Technology Integration
Leading technology integration like Artificial Intelligence/Machine Learning, Big Data and AR with medical devices
Connected App Development
Remote Diagnostics
Connectivity Support
Enabling healthcare devices with BLE, Wi-Fi, LTE/4G/5G based connectivity
Cloud-based Development
Expertise in development based on HIPAA compliant cloud-like ClearData, AWS
Tools Expertise
Expertise in tools like VectorCast, requirement traceability, & >95% code coverage
End-to-end Testing
Experience in test strategy, requirement testability, dry runs & formal execution, & risk analysis
Why VOLANSYS?
We excel at all stages of medical grade device development, cloud integration, application development, testing and large scale manufacturing